Anatomy
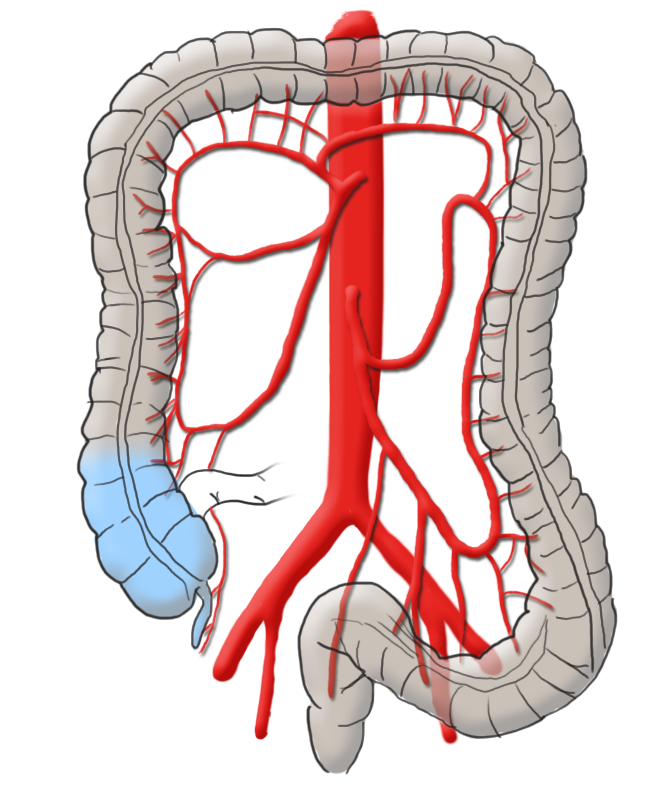
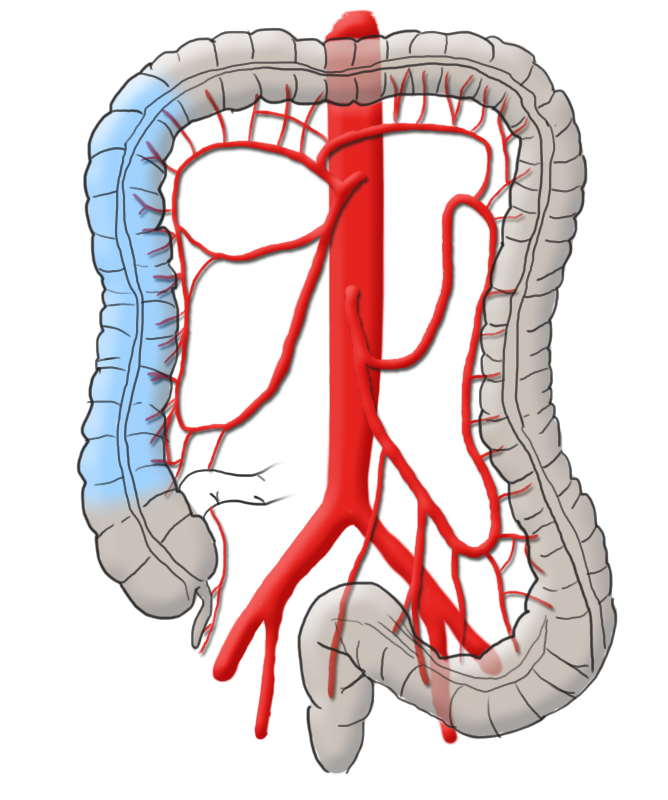
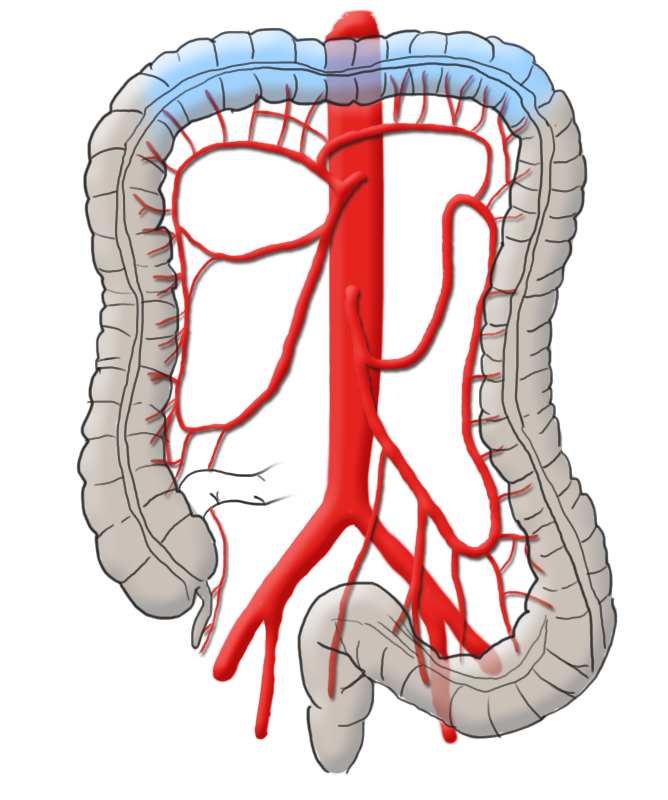
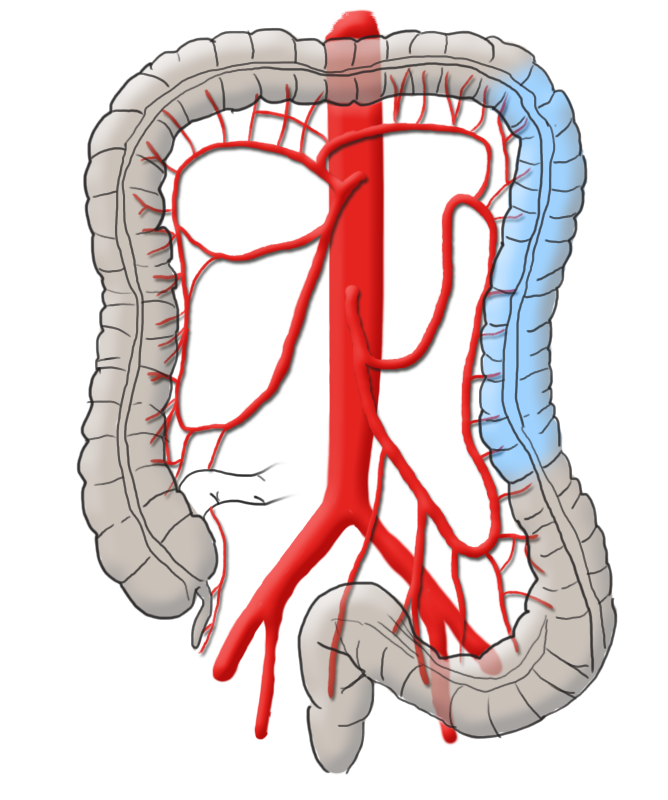
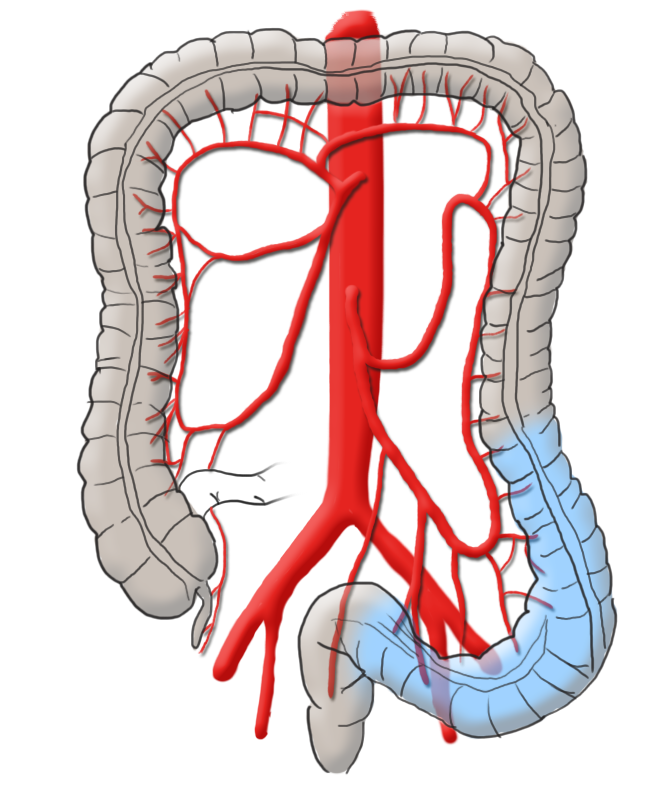
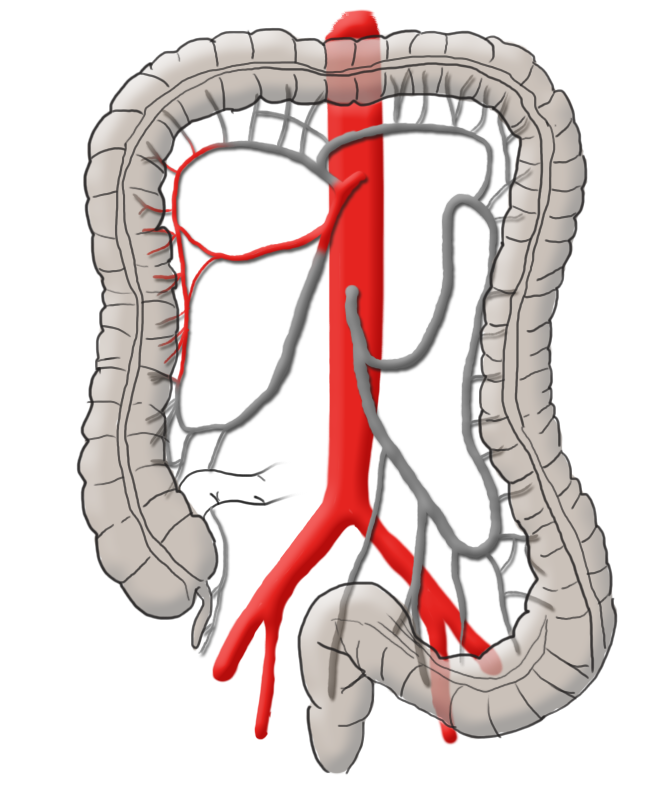
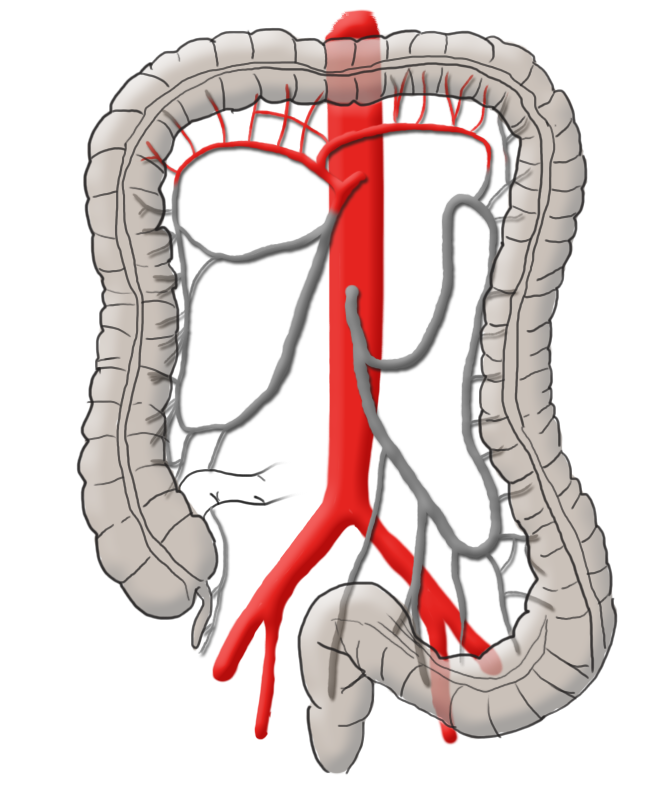
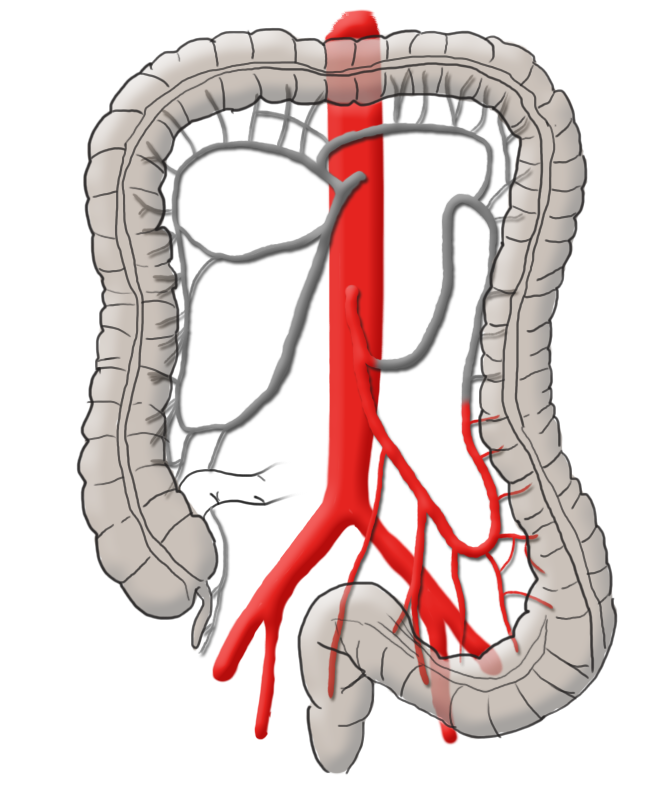
The colon is divided into five segments. It begins with the cecum communicating with the terminal ileum through the ileocecal valve (Bauhin’s valve) at the ileocecal junction. Continuing aborally is the ascending colon, which to the transverse colon at the right flexure transitions.
After the left colic flexure comes the descending colon and finally the sigmoid colon, which transitions siphon-like into the rectum. Macroscopically, this segment is characterized by the transition of the taenia of the colon into the circular muscle layer of the rectum.
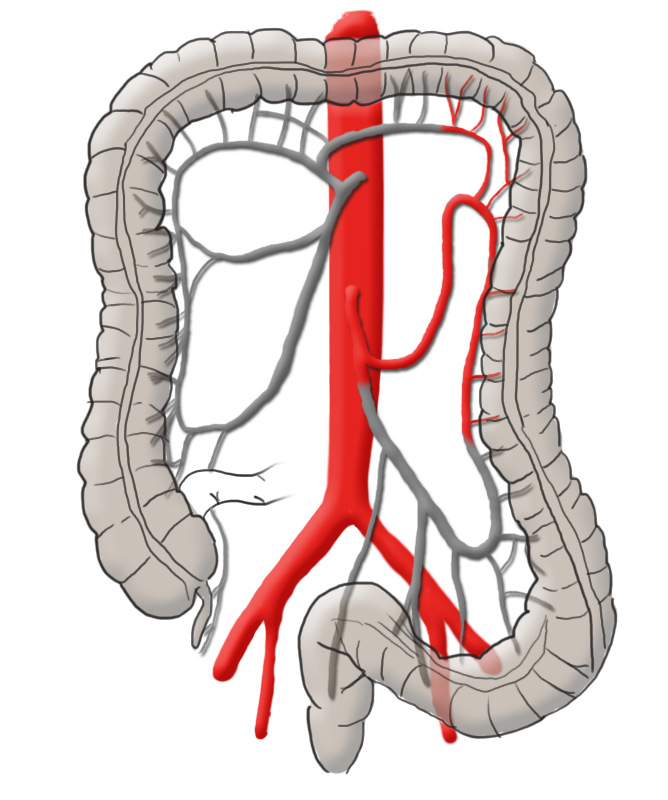
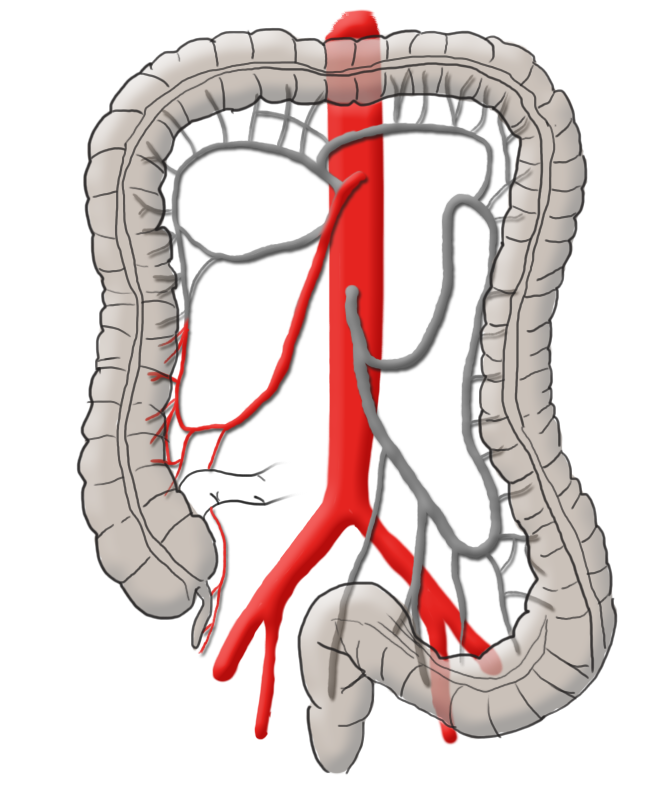
The arterial blood supply runs through the superior and inferior mesenteric arteries, which meet near the left colic flexure at the so-called anastomosis of Riolan. The right hemicolon up to the left flexure is thus supplied by the superior mesenteric artery with its lateral branches the ileocolic artery, middle colic artery, and the right colic artery. The latter in particular is in rare cases absent. The inferior mesenteric artery has the branches sigmoid artery and left colic artery and supplies a portion of the rectum.
Etiology
Colorectal carcinoma is the third most common type of cancer in Germany and the second most common cancer-related cause of death worldwide.1
The high rate of colorectal carcinoma in the Western world suggests a connection with Western dietary habits. Among the risk factors are a diet low in fiber and rich in meat and saturated fats, compounded by a sedentary lifestyle and obesity. Add to this alcohol and nicotine consumption. The classic carcinogen of colorectal carcinoma is benzopyrene, which in contained e.g. in fat dripping on charcoal and can condense on grilled foods. When consumed it can cause DNA damage, which the body routinely repairs. But if there is an imbalance or if the repair mechanisms are impaired, carcinoma can arise.
The S3 guidelines for colorectal carcinoma recommend a fiber-rich diet as well as low meat consumption, limited alcohol consumption, and a calcium and folic acid-rich diet. It also recommends regular exercise and weight loss for persons with a BMI >25kg/m².
Comprising about 95% of all colon carcinomas, the sporadic form is much more common than hereditary forms. Colon carcinomas usually arise from polyps or adenomas. In 1951 the adenoma-dysplasia-carcinoma sequence was first postulated. This sequence forms the basis for the screening practice common in Germany. After the 50th year of life, health insurance companies pay for an early detection colonoscopy every 10 years. Every polyp discovered at these exams is removed. This practice has dramatically reduced the incidence of colorectal carcinoma in Germany. Another screening procedure is the test for occult blood in stool, called the hemoccult test. If repeated three times it has a sensitivity of 30% to 90% and a specificity of 90% to 99%. This test allows early detection of asymptomatic colon carcinomas. It should be performed annually beginning at age 50 in patients who decline colonoscopy.
Flexible sigmoidoscopy is also recommended if complete colonoscopy is declined. Colorectal carcinomas are most often encountered in the distal segment. Flexible sigmoidoscopy can be combined with hemoccult tests. Radiologic screening procedures are also available, including virtual colonoscopy performed with CT or MRI. These procedures have the drawback that they may expose the patient to radiation and lack the possibility of intervention.
Precancerosis
The term precancerosis is applied to a disease or change that is highly likely to lead to the development of invasive carcinoma. A distinction is made between facultative and obligate precancerosis. A facultative precancerosis has a high probability of malignant degeneration, obligate forms always develop into carcinoma, the only question is when.
Among the facultative precanceroses are — in addition to colon adenomas – the changes that occur in hereditary nonpolyposis colorectal cancer (HNPCC). The name is somewhat confusing, it sounds as though polyps do not play a role in the development of these carcinomas, which is of course not the case. The term „nonpolyposis“ is merely meant to differentiate this disease from polyposis, in which hundreds of polyps occur. To assess the probability of an HNPCC, anamnestic criteria have been developed for its diagnosis. In the Amsterdam criteria, all of the criteria must be met to diagnose an HNPCC. If one or more of the Bethesda criteria are fulfilled, further molecular-genetic clarification should be done.
HNPCC, in which the mismatch repair genes are mutated, is associated with colorectal carcinomas, endometrial carcinomas, and gastric carcinomas. The risk of developing colon carcinoma for HNPCC patients is about 50%. An obligate precancerosis is familial adenomatous polyposis (FAP). Patients with FAP have an autosomal dominant inherited gene defect in the adenomatous polyposis coli (APC) tumor-suppressor gene and have a 100% chance of developing colorectal carcinoma. In such cases, complete removal of the colon and rectum is advised.
Special Risk Factors
Patients with ulcerative colitis have a 2 to 8-fold elevated risk of developing colon carcinoma. Unlike carcinomas that develop from polyps, colitis-associated carcinomas tend to grow superficially and are rarely polypoid or exophytic.
The earlier the disease breaks out and the longer is persists, the greater the risk of colon carcinoma. Complete inflammation of the entire large intestine, i.e. pancolitis, also increases the risk.
Patients with severe ulcerative colitis are therefore often advised to undergo a colectomy-proctomucosectomy, in which the entire large intestine with the rectum and mucosa of the anal canal are removed. The intestinal continuity (Passage) is then reconstructed with an ileo-anal pouch.
| HNPCC: AMSTERDAM CRITERIA |
|---|
| At least three members of the family have histologically confirmed colon carcinoma |
| At least one of these members is a first degree relative of the other two |
| At least two consecutive generations have colorectal cancer |
| At least one of the affected relatives was diagnosed with colorectal cancer before age 50 years |
| Exclusion of a familial adenomatous polyposis |
- For the diagnosis of HNPCC all criteria must be fulfilled
- Amsterdam II criteria: inclusion of associated non-colorectal cancers equated with colorectal carcinoma (endometrial carcinoma, small intestine cancer, hepatobiliary carcinoma in the absence of a viral genesis, transitional squamous cell carcinoma of the efferent urinary tract)
| HNPCC: BETHESDA CRITERIA |
|---|
| Two relatives first grade with colorectal cancer and/or HNPCC-associated carcinoma before age 45 years |
| Two relatives first grade with colorectal cancer and/or HNPCC-associated carcinoma before age 40 years |
| Occurrence of an „undifferentiated“ colon carcinoma in the right hemicolon before age 45 years |
| Mucinous / signet-ring cell carcinoma before age 45 years |
| Demonstration of two synchronous / metachronous colon carcinomas or other HNPCC-associated carcinomas |
| Amsterdam criteria positive |
- Even if only one of the Bethesda criteria is fulfilled, an HNPCC is considered very probable and further molecular-genetic analysis should be done
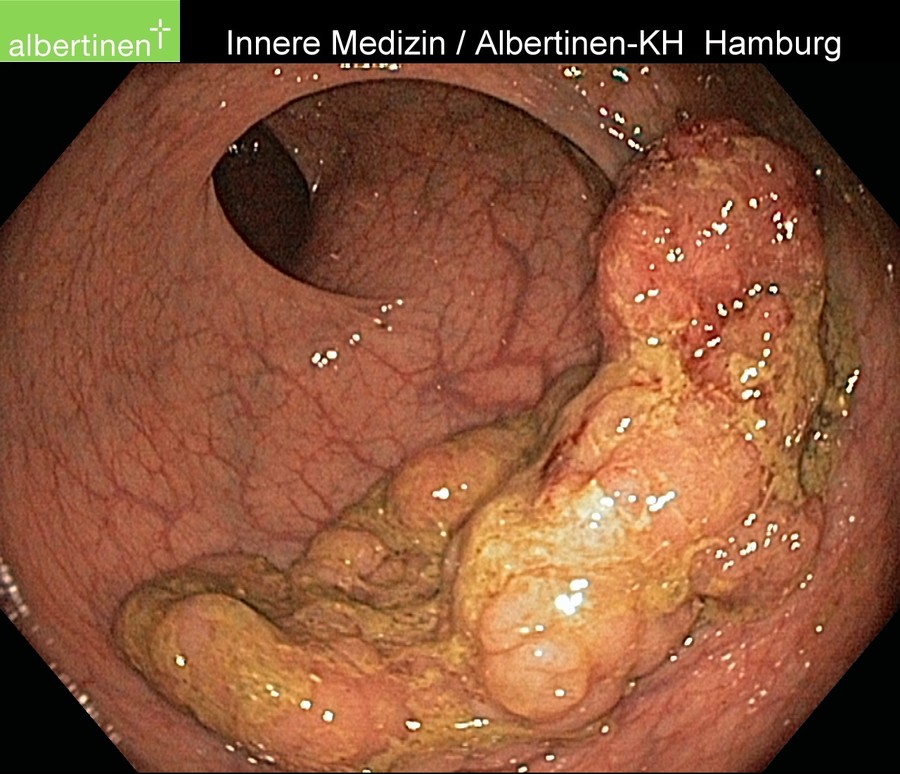
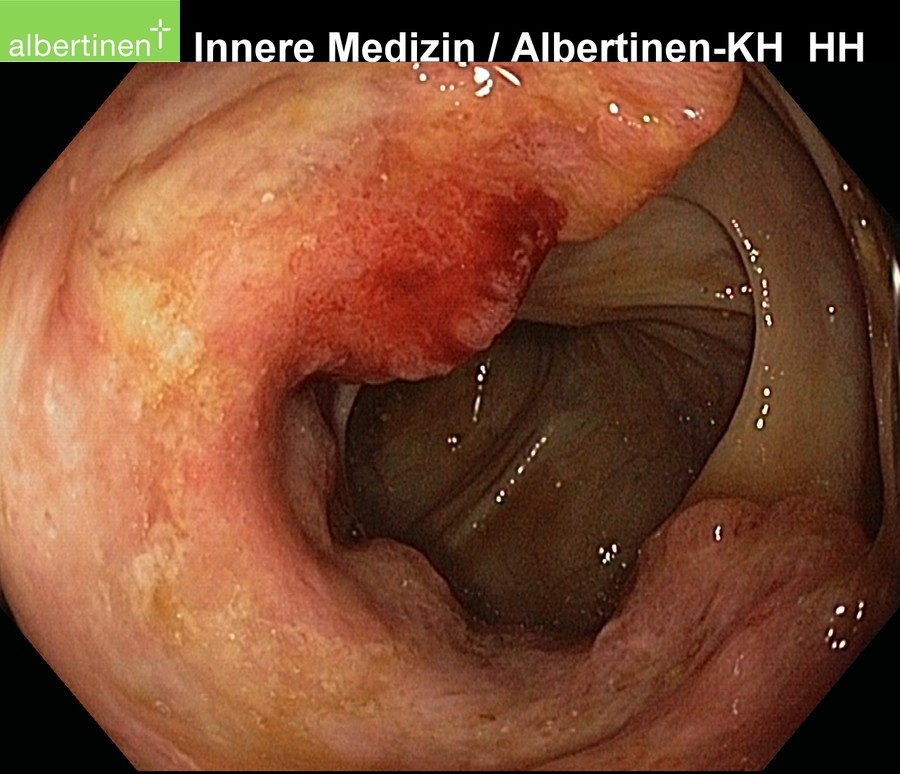
Clinical Findings & Symptoms
Every change in bowel habits should be clarified with regard to a [possible] carcinoma
The problem with colon carcinoma is that they give rise to symptoms late rather than early and when they do arise the symptoms are usually very nonspecific. Pain and abdominal colic can occur, as can fatigue and diminished capacity. Blood in the stool and a change in bowel habits are further signs. Paradoxical diarrhea is caused by stenoses and is usually a late symptom. Pencil thin stools are caused by distal stenoses, i.e. most likely by rectum carcinomas.
with courtesy by Albertinen-Krankenhaus Hamburg
Diagnosis
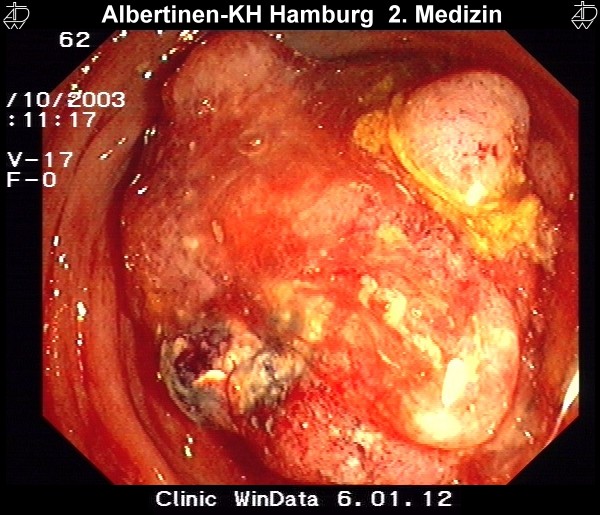
Endoscopy is the method of choice for diagnosis of colon carcinoma. Because the symptoms tend to be nonspecific and arise late, diagnosis is often made during early detection colonoscopy. The endoscopist can document the degree of stenosis of a carcinoma, the type of growth, and its approximate location. It should also always be clarified whether there is a multifocal involvement, termed a synchronous second carcinoma.
If the tumor cannot be passed with the endoscope, a complete colonoscopy should be repeated shortly after surgery, provided colon remains in the body that could not be viewed during the initial diagnostic exam. In addition, histologic confirmation and removal of additional polyps are also possible.
Endosonography plays rather a subordinate role in colon carcinoma. This is because it is technically more cumbersome to perform in the colon than in the rectum and because it would have no bearing on therapy since colon carcinoma cannot at present be given neoadjuvant therapy. This may change in future, studies are currently examining neoadjuvant therapy in colon carcinoma.
Die Endosonografie spielt beim Kolonkarzinom eher eine untergeordnete Rolle, zum Einen, weil sie technisch wesentlich aufwendiger ist als im Rektum, zum Anderen, weil sich keine wirkliche therapeutische Konsequenz daraus ergibt, da Kolonkarzinome im Gegensatz zu Tumoren im Rektum aktuell nicht neoadjuvant vorbehandelt werden. Dies könnte sich in Zukunft ändern, im Rahmen von Studien wird die neoadjuvante Therapie auch beim Kolonkarzinom überprüft.
Beispiel einer koloskopischen Polypektomie
Staging
To exclude distant metastases in a confirmed colon carcinoma, CT of the thorax and abdomen is carried out. According to the guidelines, ultrasound of the liver and x-ray of the thorax are also effective, but CT is now so cost-effective and widely available that it has become established as the standard procedure.
TNM 7 2009 – deutsche Auflage 2010
Operative Therapy
As in rectal carcinoma, removal of the tumor must include a sufficient safe resection margin. A basic part of every oncological resection of the colon is radical resection of all lymph nodes in the drainage area of the tumor. The supplying arteries are also ligated radicular, i.e. near where they branch off from the aorta, thus ensuring removal of all lymph nodes in the drainage area. The resected portion of the large intestine is longer than is necessary for the immediate safe resection margin. The perfusion status after radicular ligation of the vessels and lymphadenectomy, therefore, determines the length of the resected colon, not the immediate safe margin.
Surgical Procedure
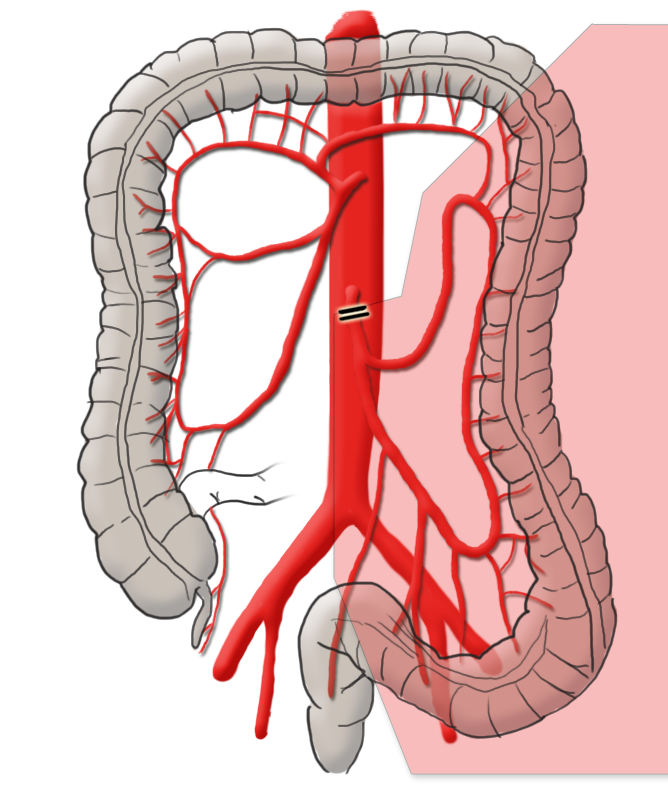
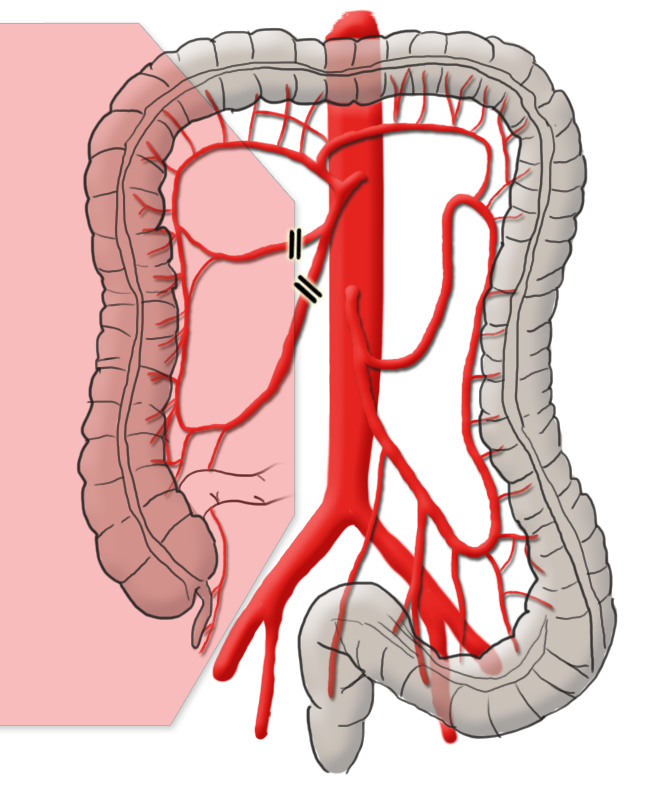
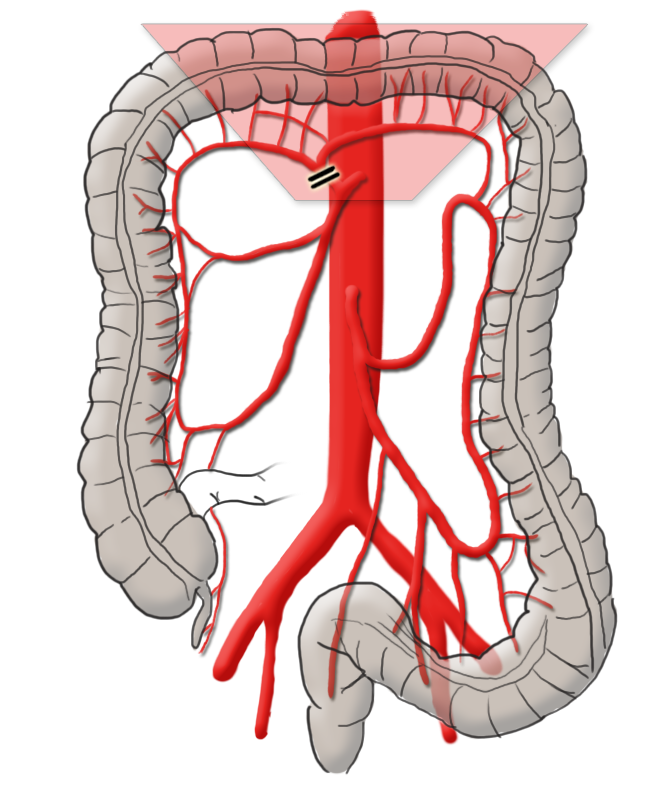
There are three basic oncological procedures for resection of the colon, right hemicolectomy, transverse colectomy, and left hemicolectomy.
In right hemicolectomy, the ileocolic artery and the right colic artery are removed at their origins, extended right hemicolectomy includes the middle colic artery as well. The intestinal continuity is reestablished with a termino-terminal ileo-transversostomy.
Tumors located exactly in the middle of the transverse colon are removed by oncological transverse resection under central ligation of the middle colic artery. If however the tumor is located near one of the flexures, extended right hemicolectomy or extended left hemicolectomy is preferred.
The perfusion status after radical lymphadenectomy determines the length of the resected colon
Tumors in the left hemicolon are radically removed by oncological left hemicolectomy. The inferior mesenteric artery is resected close to its origin and bowel continuity restored by a termino-terminal descendorectostomy in which Riolan’s anastomosis plays a special part. After ligation of the inferior mesenteric artery, the superior mesenteric artery supplies blood through Riolan’s anastomosis to the descending colon and the anastomosis of the intestine. If this is not made, the transverse colon must be carefully further mobilized to ensure sufficient perfused bowel for an anastomosis. There often then remains no other option but to perform a subtotal colectomy with counterclockwise construction of an ascendo-rectal anastomosis.
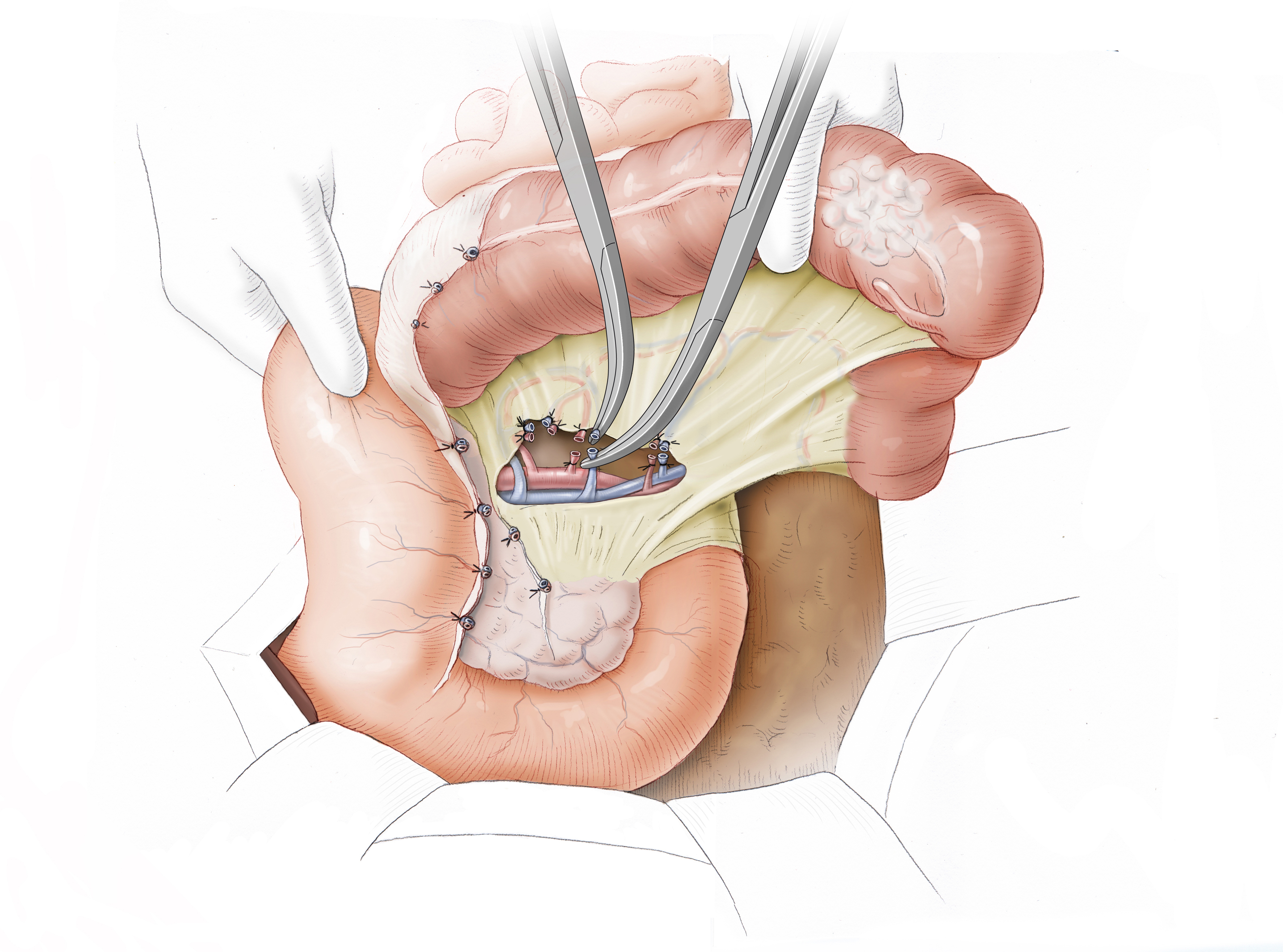
Complete Mesocolic Excision (CME)
R.J. Heald introduced the concept of complete mesorectal excision (CME) more than 20 years ago2 Now accepted as a standard procedure, it involves complete removal of the perirectal fat tissue with preservation of the fascia. (Faszienhüllen) This removes all of the locoregional lymph nodes in rectal carcinoma and greatly improves the oncological quality.
A hemicolectomy should also involve systematic lymphadenectomy with ligation at their origins of the ileocolic artery, right colic artery, and the right branches of the middle colic artery. This radical procedure involves a lymphadenectomy along the superior mesenteric artery and vein and can — like the rectal surgery — be described as a CME. The concept was first described by Hohenberger et al. in 2008.
3
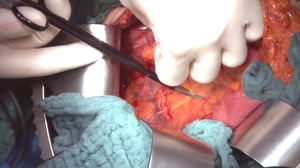
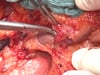
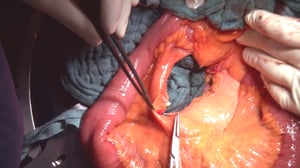
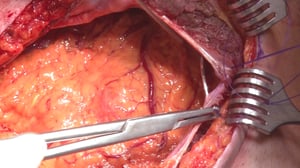
Op-Video: Right hemicolectomy with CME
Seen here is a step-by step video of an oncological right hemicolectomy with CME. The patient had a colon carcinoma located in the cecum.
1 Positioning & Overview
- Preparations for right hemicolectomy
- Positioning of the patient in a supine position.
- Upper abdominal transverse laparotomy ca. 5 cm above the navel
- Opening of the anterior rectus sheath
- Severing of the rectus abdominis muscle
- Opening of the peritoneum
2 Exploration
- The transverse colon and great omentum are lifted out
- The site is probed to exclude a peritoneal carcinomatosis
- Insertion of a ring retractor and Ulmer cable pull retractor as retractor system
3 Tumor Localization
Examination of the colon and palpation of the tumor in the cecum
4 Mobilization of the Right Hemicolon
- Detachment of embryonal adhesions of the right semicolon to the abdominal wall
- Mobilization of the right hemicolon from the paracolic gutters
- Mobilization (Auslösen) of the right colic flexure
- The colon is increasingly mobilized and lifted little by little out of the OP site by the assistants
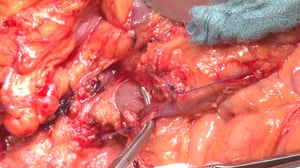
5 Preparation of the Duodenum and Mesenteric Axis
- The right hemicolon is now largely mobilized
- Underneath, the duodenum is exposed in its retroperitoneal position
- The duodenum is gradually dissected
- The head of the pancreas is exposed and carefully preserved
- The greater omentum is detached from the transverse colon as far as necessary for later reconstruction
- Often veins run directly from the mesenterial axis to the duodenum. These must be dissected and ligated
- Dissection is continued along the pancreas to expose the superior mesenteric artery and vein
- First the superior mesenteric vein is exposed
- Often the superior mesenteric artery runs around the artery
- The goal of the next step is central resection of the ileocolic artery and vein, thus achieving complete mesocolic excision with systematic lymphadenectomy
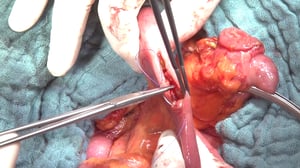
6 Central Resection of the Ileocolic Artery and Vein
- The surgeon begins by resecting (absetzen) the ileocolic artery between two Overholt ligatures. This typically runs around the superior mesenteric artery
- The more radical resection (Absetzen) of the ileocolic artery directly at its origin in the superior mesenteric artery guarantees the radical lymphadenectomy
- In the context of hemicolectomy, this procedure is also termed a complete mesocolic excision, or "CME"
- The ileocolic vein is severed and ligated in the same manner
- At the resection margins (Absetzungsrand) of the vessels a window is dissected through the mesocolon. This facilitates medial viewing during resection (Absetzen) of the mesocolon
- Now the right colic artery and vein are dissected from medial
7 Resection & Reconstruction
- The mesentery of the small intestine is severed with ligation and severing of the remaining margin vessel
- Intestinal continuity (Passage) between the small intestine and transverse colon is restored later than in side-to-end anastomosis. Because the lumens of the two bowe l segments do not match, termino-terminal reconstruction is not done, but a latero-terminal ileotransversostomy is formed
- For this purpose, the small intestine is excised in the area of the terminal ileum using a linear stapler. The stapled seam is later sewn over
- The distal resection margin on the transverse colon is determined. The mesocolon (Messkolon) is skeletonized and severed, thus also severing branches of the middle colic artery running to the right
- Intestinal clamps are placed to prevent leakage of stool. The anterior wall of the transverse colon is then opened and cleaned, only then is it completely excised
- The resected tissue encompasses the complete right hemicolon with the radicularly excised ileocolic artery and vein and middle colic artery and vein. Resection of the entire mesocolon achieves a radical lymphadenectomy
- Reconstruction by latero-terminal ileotransverse colostomy is done. For this purpose the small intestine is opened far enough antimesenterial to compensate for the lumen incongruence with the large intestine
- The anastomosis is done in hand suture technique with resorbable Vicryl-sutures in Gambé technique.) The anastomosis can also be done with a Stapler
- Successful healing requires a tension-free, non-twisted anastomosis well supplied with blood
8 Closing the Mesocolon Window
- Reconstruction creates a window in the mesocolon through which the small intestine could herniate
- Such an interior hernia can be avoided by closing the mesocolon incision. This too is done with Vicryl
9 Closing the Abdominal Wall
- The greater omentum is put back in place
- Layered reconstruction of the abdominal wall
- First the peritoneum is closed
- Then the anterior rectus sheath is sutured closed (not shown in video)
- Followed by skin closure using Donati back-and-forth suture technique
- Placement of an epifascial Redon drain. For a right hemicolectomy, no intraabdominal drain is placed
Follow-up treatment
The final tumor stage is determined by the pathologist and then given the prefix
„p“. The specimen should contain at least 12 lymph nodes to allow a finding regarding the N status. Colon carcinomas in UICC stage III receive adjuvant chemotherapy, for which various chemotherapy schemes and monoclonal antibodies are available.
References
- Cancer statistics, 2010.CA Cancer J Clin. 2010 Sep-Oct;60(5):277-300. doi: 10.3322/caac.20073. Epub 2010 Jul 7.Jemal A1, Siegel R, Xu J, Ward E.
- The ‘Holy Plane’ of rectal surgery.J R Soc Med. 1988 Sep;81(9):503-8.Heald RJ1
- Standardized surgery for colonic cancer: complete mesocolic excision and central ligation – technical notes and outcomeColorectal Dis. 2009 May;11(4):354-64; discussion 364-5. doi: 10.1111/j.1463-1318.2008.01735.x. Epub 2009 Nov 5.Hohenberger W1, Weber K, Matzel K, Papadopoulos T, Merkel S.


 Wound Healing
Wound Healing Infection
Infection Acute Abdomen
Acute Abdomen Abdominal trauma
Abdominal trauma Ileus
Ileus Hernia
Hernia Benign Struma
Benign Struma Thyroid Carcinoma
Thyroid Carcinoma Hyperparathyroidism
Hyperparathyroidism Hyperthyreosis
Hyperthyreosis Adrenal Gland Tumors
Adrenal Gland Tumors Achalasia
Achalasia Esophageal Carcinoma
Esophageal Carcinoma Esophageal Diverticulum
Esophageal Diverticulum Esophageal Perforation
Esophageal Perforation Corrosive Esophagitis
Corrosive Esophagitis Gastric Carcinoma
Gastric Carcinoma Peptic Ulcer Disease
Peptic Ulcer Disease GERD
GERD Bariatric Surgery
Bariatric Surgery CIBD
CIBD Divertikulitis
Divertikulitis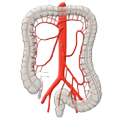 Colon Carcinoma
Colon Carcinoma Proktology
Proktology Rectal Carcinoma
Rectal Carcinoma Anatomy
Anatomy Ikterus
Ikterus Cholezystolithiais
Cholezystolithiais Benign Liver Lesions
Benign Liver Lesions Malignant Liver Leasions
Malignant Liver Leasions Pancreatitis
Pancreatitis Pancreatic carcinoma
Pancreatic carcinoma